If basic research drives innovation, the CRISPR-Cas system is one trophy-laden driver. Not long after its unexpected discovery as an obscure prokaryotic defense system against viruses, it has become the hottest thing since recombinant DNA. However, CRISPR’s intricate details still remain somewhat mysterious.
Not to fear: basic research into CRISPR rolls on, with new, intriguing, and potentially useful details coming out about as often as U.S. presidential debates. Three recent CRISPR science stories reveal even more epicycles in the bacteria-virus fight, highlighting how viruses fight back against CRISPR, how viruses fight viruses with CRISPR-like immunity, and how some CRISPR systems can acquire immunity from RNA sequences, instead of DNA.
Phages Fight Back
The Red Queen view of evolution says it’s never safe just to sit back on your laurels – you always have to keep adapting to avoid extinction. This seems to be true for viruses faced with anti-viral CRISPR systems – not only does their DNA evolve to evade CRISPR defenses, but some viruses even have weapons to take out the CRISPR guns.
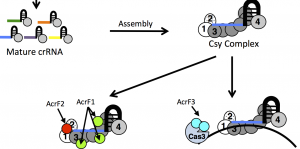
Three anti-CRISPR proteins – AcrF1, 2, and 3 – block Type I CRISPR-Cas systems in different ways. AcrF1 and 2 prevent the Csy complex from binding DNA, while AcrF3 holds off the Cas3 nuclease. Image via Karen Maxwell.
Karen Maxwell at University of Toronto laid out the battlefield conditions recently in PLoS Pathogens. While looking at a family of phages that infect Pseudomonas aeruginosa bacteria, her lab found a region containing anti-CRISPR genes. This locus was conserved across phages, but the actual genes varied from phage to phage, with some taking out Type I-E CRISPR systems, and others focusing on Type I-F. Three tested anti-CRISPR proteins each worked in a slightly different way, but all blocked the full Type I Csy-Cas complex from cutting target DNA.
Unfortunately for evolutionary biologists out there, the anti-CRISPR proteins don’t have much homology to known sequences, and there aren’t enough of them known to do much phylogenetics. However, where there are nine anti-CRISPR systems, there are likely to be hundreds; we just haven’t found them yet.
Interestingly, anti-CRISPR protein AcrF3, which allows Csy to bind DNA, but prevents Cas3 from cutting, converts the CRISPR system into a transcriptional repressor. This could be another useful tool for synthetic gene regulation or epigenome editing using CRISPR, adding a third technique in addition to catalytically dead Cas9 and imperfect guide RNAs. It could also potentially be useful for controlling or delaying CRISPR gene edits, or dynamically switching between gene editing and repression.
Virus Vs. Virus
One of the more intriguing stories of the past decade is giant viruses. If CRISPR blurred some category boundaries by showing bacteria have adaptive immunity, giant viruses are busting through these walls with more gusto than the Kool-Aid man. Not only are giant viruses big, but they have big genomes, and they are even plagued by virus-infecting viruses (virophages) themselves!
Inspired by anti-viral CRISPRs in prokaryotes, Anthony Levasseur and Meriem Bekliz went fishing for anti-virophage systems in giant mimiviruses. In not too long, they caught a suspiciously CRISPR-like array of four 15 bp repeats from an offending virophage genome. The first of these repeats was extended to include 28 bp of virophage DNA, and the whole array was integrated inside one of the potentially important genes.
The team called the array a Mimivirus virophage resistance element, or MIMIVIRE. It’s not clear how it works, or if all four repeats are necessary, but silencing any of the three putative defense genes blocked the mimivirus’s defense against its virophage. We still don’t know much about how MIMIVIREs work, but they may someday be a contender in the quest for the best CRISPR system for genome editing.
Acquiring CRISPR Immunity From RNA
Of the many CRISPR sub-types, some target DNA, and some target RNA. Targeting RNA would be important for defense against RNA viruses, but that still doesn’t explain how cells would acquire anti-RNA immunity in the first place, since we’ve only seen CRISPR systems acquire targeting spacers from DNA, not RNA.
Concerned by this conundrum, Sukrit Silas and Georg Mohr noticed that some Cas1 genes, which integrate CRISPR spacers, are fused to reverse transcriptases, which create DNA copies of RNA. Hypothesizing that this combo would help cells acquire spacers from RNA, they overexpressed the natural RT-Cas1 in Marinomonas mediterranea and looked for new spacer acquisition from endogenous and plasmid-borne genes.
Indeed, new CRISPR spacers were proportional to the RNA expression level of each gene, an effect that disappeared if the reverse transcriptase was broken. With a clever experiment using a self-splicing intron, they made a highly expressed RNA sequence not present in DNA, and showed the system could acquire that sequence directly from RNA.
We don’t yet know where these new discoveries will lead, but they are yet more proof that the world out there always holds more secrets than we realize. To see what you might use these discoveries for, check out this latest crop of basic CRISPR research in PLoS Pathogens, 2016; Nature, 2016; and Science, 2016



